Malaria
Although malaria is no longer endemic in Italy, it still represents the most important imported disease in our country, due to the growing number of travelers from endemic areas.
CAUSATIVE AGENT AND INFECTIVE CYCLE
The name malaria comes from the ancient belief that this disease could be contracted from the "bed air" ("mala aria" in Italian) i.e., the unhealthy miasmas emanating from the stagnant water of the marshes. In this belief, there was some truth, namely the close link between the disease and the marshy areas. However, today we know that the correlation between the two is found in a couple of living organisms: the mosquito and the plasmodium.
When we speak generically about malaria, we refer to different pathologies, similar yet different in symptoms, severity, and causative agents. The latter are represented by several species of parasitic Protozoa of the Sporozoan class, all of which belong to the genus Plasmodium and can cause one or more of the peculiar forms of the disease. There are four common pathogenic plasmodia for humans: Plasmodium falciparum, the agent of the so-called malignant tertian (the most severe form), P. vivax and P. ovale, agents of two forms of benign tertian, and P. malariae, the agent of quartan.
A fifth Plasmodium, P. knowlesi, uses primates as its reservoir for the disease in the forests of Borneo and Malaysia; despite happening rarely, it can cause a benign form of “daily” malaria also in humans. However, the disease reservoir of the four most common plasmodia is in human populations in which the disease is chronic.
There are about sixty species of mosquitoes with nocturnal crepuscular activity (even if there are only 3 - 4 species capable of transmitting the disease in each geographical area), which all belong to the genus Anopheles. Mosquitoes that belong to other genera are not able to transmit these parasites (while they can transmit viruses, bacteria, and microfilariae).
Not all Anopheles can act as a vector. For this to happen, there must be perfect temporal concordance between the so-called gonotrophic cycle of the mosquito (blood meal, maturation and egg laying, new blood meal) and the development of plasmodium in the mosquito itself.
It is also necessary that the mosquito has a high anthropophilic index, which means that it is highly probable that its next blood meal will be on a human (e.g., An. Gambiae, one of the main African vectors has an anthropophilic index from 80 to 100%).
If a mosquito can be a potential vector, for it to trigger an epidemic cycle, it must be present in sufficient quantities and there must also be enough infected human subjects from which to obtain “supplies” of plasmodia. These conditions do not occur anymore in many places around the globe, this includes Europe, much of Asia, and North America, where these species remain in areas of so-called "residual anophelism".
The introduction of plasmodia through travelers returning from endemic areas can cause the appearance of sporadic autochthonous cases; this only happens if the mosquito is competent for the introduced plasmodium by the traveler. Malaria is only contracted after an infected female’s bite and provided that she belongs to the genus that is competent for the transmission of the plasmodium.
In turn, infection will only be transmitted to a mosquito by sucking the blood from a malarial subject in the infectious phase and before it can infect a healthy subject, the plasmodium must complete a part of its developmental cycle within the mosquito itself. This cycle can last from a week to almost a month depending on the plasmodial species and the ambient temperature.
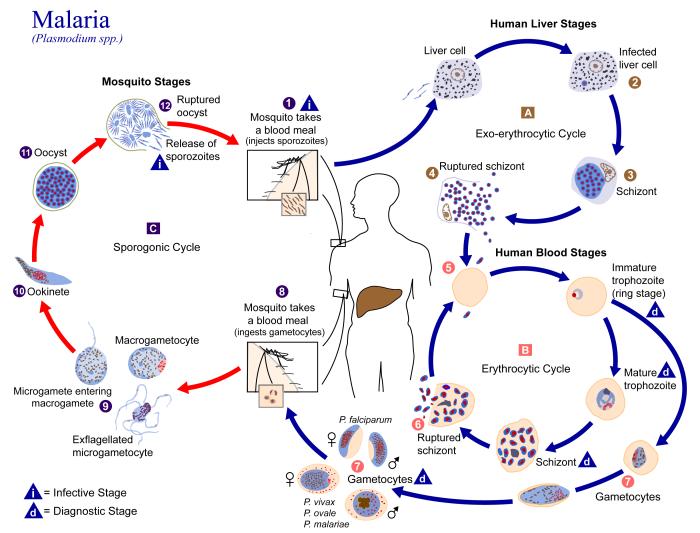
The biological cycle of malaria.
Source: CDC.
When a mosquito bites a human, it inoculates the plasmodia with its own saliva at the terminal stage of evolution in the mosquito; these are called sporozoites. Within an hour of the bite, the sporozoites infect the liver cells (hepatocytes) where they multiply and establish a cycle called the exoerythrocytic stage, transforming first into trophozoites and then into schizonts (polynuclear cells).
This cycle in the liver lasts between 6 and 16 days, depending on the plasmodium species. Each schizont then bursts and releases from 10,000 to 30,000 merozoites into the bloodstream where they then go on to infect the red blood cells; consequently, the erythrocyte cycle begins. A few hours after the invasion, the plasmodia are visible under the microscope with their characteristic signet ring shape, which represents an immature trophozoite.
Towards the end of the annular stage, a digestive vacuole begins to appear. Inside it are exceptionally fine granules of hemozoin, which is the terminal pigment derived from the hemoglobin of the red blood cells on which the plasmodia feed. The digestive vacuole and the quantity of pigment increase with the growth of the parasite, known at this stage as trophozoite. After successive nuclear divisions, the digestive vacuole finds itself at the center of a new schizont with merozoites around it, taking on the form of a rosette.
At this point, the red blood cells break, releasing merozoites and pyrogenic substances into the blood that cause the first febrile state. Some merozoites infect other red blood cells, cyclically repeating the process and inducing febrile attacks. Other merozoites are transformed into male and female sexual elements called gametocytes.
If at this point a competent mosquito bites the infected host, it can ingest the gametocytes in its blood. Once this takes place, the male gametocytes will multiply several times and form flagellated microgametes, which are mobile. In contrast, female gametocytes, known as macrogametes, are larger and immobile. The fusion of the two gametes results in the formation of a zygote that soon changes shape to become a motile ookinete, which is elongated and mobile.
This can penetrate the midgut wall to form an oocyst. The oocyst enlarges over time and contains thousands of sporozoites. It then bursts to release sporozoites that migrate to the mosquito salivary gland, rendering the mosquito infectious to human beings. Once having found a new host, the cycle repeats itself.
In the case of P. falciparum, the erythrocyte cycle begins about a week after the start of the hepatic cycle and happens every 48 hours. The cycle induces recurrent febrile fits that occur every third day (malignant tertian malaria); these are similar to the malaria caused by the P. vivax and P. ovale, however the infection is less severe (benign tertian malaria). With regards to the P. malariae, the erythrocyte cycle begins after about two weeks, and endures a period of 72 hours with malarial attacks taking place every fourth day (quartan malaria). P. knowlesi has a shorter cycle of just 24 hours (daily malaria). In P. vivax infections, dormant forms, called hypnozoites, remain in the liver, and can trigger relapses even after years to come.
In contrast, P. malariae can cause recrudescence due to parasite infections that remain vital in the bloodstream. These forms evade the immune defenses and create a new malarial access when these defenses are lowered.
SYMPTOMS AND COURSE OF THE DISEASE
The disease can take on different forms depending on the plasmodium involved, the condition, and age of the affected subject. Symptoms appear after the hepatic phase, which is usually asymptomatic, when the parasites complete the first cycle of asexual reproduction in red blood cells.
The malarial attack initially starts with cycles of chills, fever, and profuse sweating, which occurs every one, two or three days, depending on the plasmodia species. In advanced stages, the fever may become more irregular or develop into a continuous state. Nausea, vomiting, diarrhea, and abdominal pain can all accompany febrile states.
In adults living in endemic areas, with adequate innate or acquired immunity, the infection can be asymptomatic, or easily treatable. In this case, it is an uncomplicated malaria. In non-immune individuals, in children and in pregnant women, P. falciparum, and sometimes P. vivax malaria, can be extremely serious and possibly fatal, with severe anemia or brain infections.
Hemolytic anemia can be profoundly serious and is caused by the loss of red blood cells. This happens both by lysis of the infected red blood cells and by the accelerated senescence of the uninfected ones, which are eliminated in the spleen. Any transfused ones are quickly destroyed. The spleen is often swollen and because it contains many parasites, it can become so fragile that it can break.
Another characteristic of the P. falciparum malaria is cytoadherence. The infected red blood cells adhere to the capillary walls of various organs, including the lungs, kidney, and brain; this causes blood flow to slow down and occlusions that eventually lead to ischemia. These happen to be severe in the brain (cerebral malaria) with loss of consciousness, cerebral edema, coma and often death.
In sub-Saharan Africa, mortality from cerebral malaria can reach 20% in children under 5 years of age, even if treated pharmacologically. It is fatal in most untreated cases. In addition to cytoadherence, P. falciparum malaria is more serious because red blood cells of all ages are infected. However, P. vivax and P. ovale mainly infect young reticulocytes, while P. malariae infects senescent red blood cells.
In these less severe forms of malaria, mortality is often due to the collapse of the spleen or kidney damage. Nevertheless, recent, and more accurate diagnoses have revealed a higher number of severe P. vivax malaria than previously thought.
P. knowlesi exhibits a 24-hour schizogonic blood cycle, typically causing an uncomplicated and sensitive form to chloroquine treatment. However, several severe forms have been described in recent literature, six of which have a fatal outcome.
TREATMENT AND PREVENTION
For many years, the only known remedy used effectively for the treatment of malaria was Cinchona officinalis bark extract. The plant was used in Peru in traditional medicine and imported to Europe by the Jesuits in the early 17th century. In the 19th century, two French chemists, Pellitier and Caventou, managed to extract the active ingredient, which was an alkaloid called quinine, from the bark. It has been synthesized since 1929.
Despite its severe side effects, quinine is still used for the treatment of cerebral malaria. Due to the reduced possibility of importing Cinchona officinalis bark into Europe during the First World War, previous studies of an alternative treatment were resumed. In 1891, Paul Erlich conducted studies on the antimalarial activity of methylene blue and its chemical structure that seemed to synthesize new antimalarial substances, which included pamaquine, primaquine, and mepacrine.
In the 1940s, to protect the US troops in the Pacific, who were engaged in another war, chloroquine, amodiaquine, proguanil, and pyrimethamine were synthesized. Due to its effectiveness and low cost of production, chloroquine had a widespread use until around 1970, when resistant strains of Plasmodium began to emerge and spread.
War instigated new findings in medicine, and this time round, it was the Vietnam war. This war led American scientists to develop new compounds by modifying the structure of quinine (thus mefloquine was developed). Chinese colleagues also took to studying the products in their traditional medicine; they found the new active ingredient artemisinin in the Artemisia annua extract. Artemisinin is chemically different from other antimalarials in use, it is highly effective and safe. With regards to its chemical structure, new active principles were developed, such as artesunate, artemether, and artemotil (also known as b-arteether).
There are drugs based on aryl amino alcohols currently available (quinine, mefloquine, lumefantrine), aminoquinolines (chloroquine, amodiaquine), sulfonamides (sulfadoxine pyrimethamine or S/P), some antibiotics (doxycycline), derivatives of artemisinin (dihydroartemisinin, artemether, artemotil, artesunate, artemisone), and atovaquone, which is used in combination with proguanil as a prophylaxis under the name of Malarone®.
The pharmacological activity of artemisinin derivatives acts especially against the asexual intra erythrocyte forms of all plasmodium species. They are the only schizonticides that kill young forms of the so-called “rings” in addition with the gametocyte and its first stages of development.
Artemisinin derivatives act much faster than any other known anti-malarial drug. However, they have a noticeably short half-life that does not counteract any relapses at 3-4 weeks. For this reason, they are used in combination with other drugs in the so-called artemisinin-based combination therapies (ACT).
The treatment of uncomplicated P. falciparum malaria involves eradicating the parasite as quickly as possible to prevent the disease from evolving into a severe form, and from the development of sexual forms in humans (i.e., infectious ones). To do this, WHO recommends Artemisinin-based combination therapy (ACT) to increase the efficacy of artemisinin and reduce the onset of resistance (unfortunately already underway, even with ATC).
As an example, there is medicine on the market based on artemether and lumefantrine (Coartem® and Riamet®), artesunate and amodiaquine, artesunate and mefloquine, artesunate and S/P, or dihydroartemisinin and piperaquine (Eurartesin®).
WHO recommends starting therapy only after a confirmed diagnosis of malaria; not to use artemisinin derivatives alone; and to complete the three days of therapy. In endemic areas, especially those with a low transmission rate, it is recommended to add a single dose of primaquine as an anti-gametocyte, which can block the transmission of plasmodium to the vector. Chloroquine can be used for P. vivax infections due to the parasite still being susceptible to this drug, conversely ACT is also used in this case. This treatment eliminates the hypnozoites dormant in the liver, however, requires a two-week treatment with primaquine.
With regards to severe P. falciparum malaria, the main aim of treatment is saving the patient, and when it comes to cerebral malaria, preventing the neurological damage often associated with it. To do this, it should be treated as soon as possible with any effective medication available. In these cases, artesunate IV has been shown to be superior to quinine in both Asia and Africa but is not registered in Europe.
An antimalarial prophylactic treatment is strongly recommended for those who have come from non-endemic areas; must stay for short periods in endemic areas for tourism, work, or for volunteer work. The treatment is advised when there is a risk of contracting the severe form of the disease due to not being covered by innate or adaptive immunity.
Even if there is no pharmacological prophylaxis capable of guaranteeing 100% protection, together with your doctor, or a travel medicine specialist, it is necessary to evaluate which product to take. The destination, your state of health, your body’s tolerance to active ingredients, etc. are factors which should be assessed. The most recommended drugs are mefloquine, doxycycline, and Malarone®.
Mefloquine (Lariam®) is a fast-acting schizonticide drug, which also affects the gametocytes of P. vivax and P. malariae (but not those of P. falciparum). The use of mefloquine can cause side effects, such as digestive and central nervous system disorders. The previously mentioned association between atovaquone and proguanil (Malarone®) is also used for prophylaxis. It has a powerful schizonticidal action and is also active against the hepatic forms of P. falciparum (but not against P. vivax hypnozoites) and therefore should be taken up to a week after returning home.
Another cornerstone for preventing infection for travelers and residents in high-risk countries is protection from the vectors. Adequate protection against insect bites reduces the risk of contracting malaria by 90%. In most cases, malaria mosquitoes tend to bite from dusk when the intensity of the sun’s rays is reduced.
It is especially in these moments that it is essential that travelers take all measures possible to prevent contact with the insect:
- if possible, avoid staying outdoors when Anopheles mosquitoes are active;
- wear light-colored clothing (dark and bright colors attract insects), with long sleeves and long trousers that cover most of the body, especially after sunset;
- use insect repellent; repellents can be applied to exposed skin (or clothing) and should contain DEET (diethyltoluamide)or IR3535 or Icardin, which are the most common and effective biologically active ingredients; repellents must be used in strict accordance with the instructions on the label;
- preferably stay in rooms with an air conditioner and/or with mosquito nets on doors and windows; make sure that they are in good condition and closed well;
- if you spend the night in environments without protection, we recommend the use of bed mosquito nets; tuck the edges under the mattress and check that no mosquitoes have remained trapped inside. If necessary, it may be useful to impregnate the mosquito nets with pyrethroid -based insecticides.
Despite having taken all the precautions and that you suspect that you have contracted malaria (or other diseases) during a trip to a country at risk, you should immediately contact a center that has the possibility and ability to make an accurate diagnosis.
Many years have been dedicated to studying vaccines against the infection, but nothing has yet been found. The first vaccine to pass all the preliminary tests is the RTS,S/AS01, which has shown partial protection against P. falciparum malaria in children.
In large scale clinical trials, the vaccine was able to prevent about 4 in 10 cases of malaria; this was carried out over a 4-year period among 5 to 17-month-old infants, who received 4 doses.
WHO began a larger-scale test in April 2019 that involves the gradual introduction of RTS,S/AS01 into the vaccination program of 3 Sub-Saharan African countries (Malawi, Ghana, and Kenya). This will allow 360,000 children a year to be vaccinated. The test will help us understand the efficacy of the vaccine; the best way in which the 4 doses will be administered; its safety in routine use; and the potential role it may have in reducing infant deaths.
Another important form of prevention is the fight against the vector. The interventions to reduce the number of mosquitoes, are carried out through treatments with residual action insecticides inside houses. The treatments ensure the reduction of larval outbreaks through environmental remediation and with the use of larvicidal products, or natural predators of mosquito larvae.
Resistance to the most used insecticides has long been a cause of great concern for world health authorities. To date, over 68 countries have reported resistance to one of more classes of insecticides commonly used to impregnate bed nets, or residual treatments in homes. Until new products are introduced onto the market, WHO stresses the need to develop effective insecticide resistance management strategies and take on in the most effective approach against resistance, which is the rotation of existing active ingredients.

Cinchona officinalis.
Source: Icones plantarum medico-oeconomico-technologicarum cum earum fructus ususque descriptione, [1800] - 1822.
IMPACT OF THE DISEASE
Adult populations living in areas of high transmission develop a sort of partial or adaptive immunity to the plasmodium to which they are exposed. This form of immunity does not eliminate the risk of getting sick, but it reduces the possibility of falling ill and developing a more severe form. In these areas, the greatest number of deaths is recorded among children under the age of 5.
Only those who manage to survive past this young age can develop the adaptive form of plasmodium immunity. The second risk category is pregnant women, due to them being more susceptible to acute forms of the disease with severe anemia. This can severely impair fetal growth and lead to the death of the mother and/or unborn child.
In places where the transmission intensity of plasmodium is low, the whole population has the same risk of developing severe form of the disease.
Extreme phenomena in some areas of the third world such as floods, famines, revolutions, genocides, drastic environmental changes, etc. often cause large migratory movements to new areas. However, these can favor exposure to a parasite of populations vulnerable to it. Since these areas are generally poor, there is usually a lack of effective vector control activities and adequate infrastructures to deal with and cure the disease; all this leads to serious epidemics in a noticeably short amount of time.
In addition, malaria has a significantly negative impact on the economy. In countries where it is widespread, where the more severe forms are not contracted due to adaptive immunity, broad sections of the population are still affected and are unable to work effectively for long periods. High healthcare costs must also be covered for the prevention and treatment of the disease.
Last modified: Mar 2024



